Site pages
Current course
Participants
General
MODULE 1. Magnetism
MODULE 2. Particle Physics
MODULE 3. Modern Physics
MODULE 4. Semicoductor Physics
MODULE 5. Superconductivty
MODULE 6. Optics
LESSON 26. Types of LASER
Steady State Equilibrium
Absorption transition, spontaneous transition and stimulated transition processes are happened simultaneously. However, under steady state condition the absorption process and emission process balance each other. Therefore, theoretically
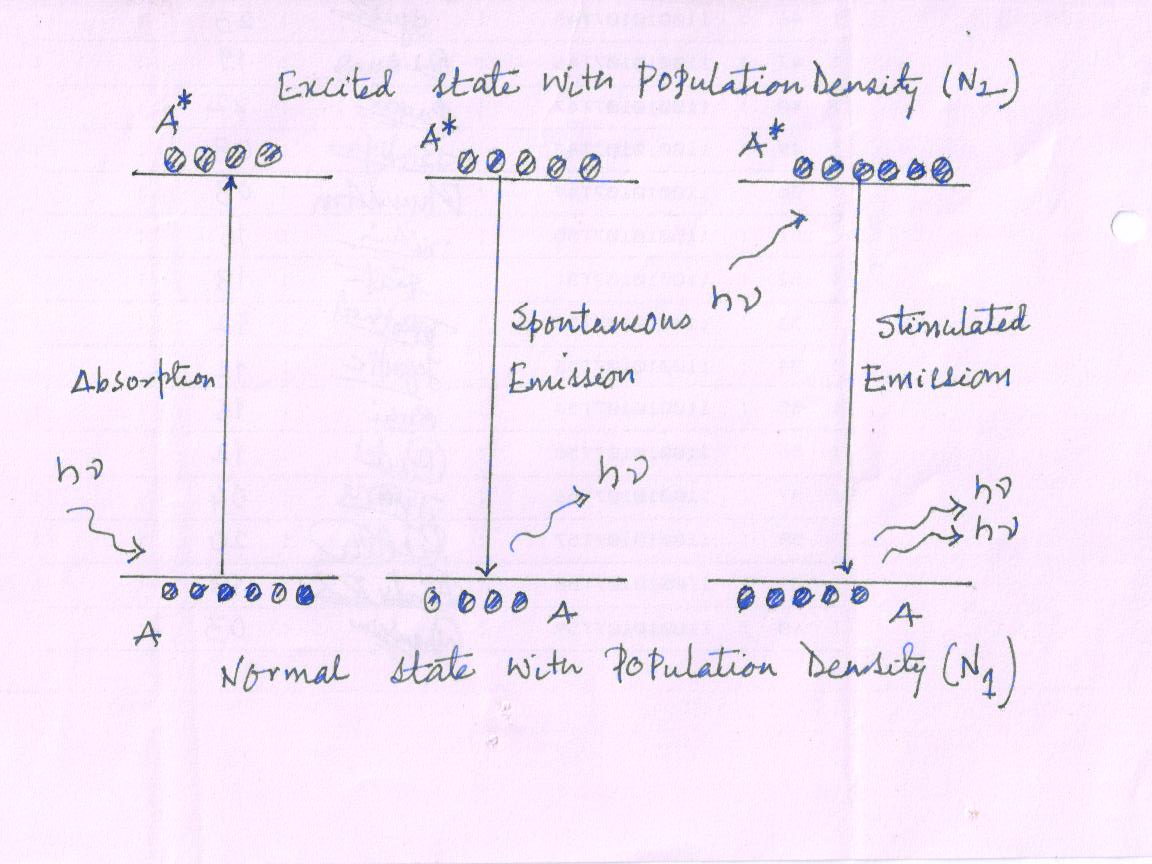
Fig17(1)
Rate of Absorption transition = Rate of spontaneous transition + Rate of stimulated transition
R12(absortion) = R21(spontaneous) + R21(stimulated)
B12N1ρ(ν)Δt = A21 N2 Δt + B21 ρ(ν) N2 Δt
At thermal equilibrium condition,
Number of atoms in the lower energy state (N1 ) are more than higher energy state (N2)
Hence, N1 >> N2
Photon traveling through medium is more like to get absorbed compare to stimulate to an excited atom to emit a photon (stimulated emission). An atom that is at the excited state is more likely to jump the lower level on its own (spontaneous) way than (compare to) being stimulated by an external photon. Because of the external incident photon density is not sufficient to interact with the excited atom. So, the spontaneous emission dominates the stimulated emission. So, at steady state condition large population is available at lower energy state. As probability for absorption transition is equal to the probability for stimulated transition.
Population Inversion
At thermal equilibrium, population ratio is governed by the Boltzmann factor according to equation
\[{{{N_2}} \over {{N_1}}}\] = \[{e^{ - {{h\nu } \over {kT}}}}\] OR \[{{{N_1}} \over {{N_2}}}\] = \[{e^{{{h\nu } \over {kT}}}}\]
hν = E2 - E1
N2 - Number of atoms in the higher energy state
N1 - Number of atom in lower energy state.
k - Boltzmann Factor
t – Temperature
It reports the population N2 at the excited level E2 will be for smaller than the population N1 at the lower level E1 hence; N1 >> N2
The condition in which there are more atoms in lower energy state and relatively lesser number of atoms in higher state is called normal state or state of equilibrium.

Fig17(2)
The condition of material in which the population of the upper energy level (N2) far exceeds the population of the lower energy level (N1).
In this situation, the population distribution between the level E1 and E2 is inverted and hence it is called as state the inverted state of non-equilibrium.
Invert state or non-equilibrium state only for a short time.
Population inversion is obtained by employing pumping technique, which transfer large number of atoms from lower energy state to higher energy
He-Ne Laser
-
A Helium-Neon laser or He-Ne laser is a type of gas laser.
-
The gain medium of He-Ne Laser consists of a mixture of helium and neon inside of a small bore capillary tube, usually excited by a DC electrical discharge.
Construction and operation
-
The gain medium of the laser, a mixture of Helium and Neon gases, in approximately a 10:1 ratio, contained at low pressure in a glass envelope.
-
The gas mixture is mostly helium, so that helium atoms can be excited and it collides with neon atoms, exciting some of them to the state that radiates 632.8nm .
-
Without helium, the neon atoms would be excited mostly to lower excited states responsible for non-laser lines. A neon laser with no helium can be constructed but it is much more difficult without this means of energy coupling.
-
The He-Ne laser that has lost enough of its helium (e.g., due to diffusion through the seals or glass) will most likely not lase at all since the pumping efficiency will be too low
-
The He-Ne lasers are relatively small devices having cavity lengths usually ranging from 15cm to 50cm . But sometimes up to about 1 meter to achieve the highest powers and optical output power levels ranging from 0.5 to 50mW .
-
The energy or pump source of the laser is provided by a high voltage electrical discharge passed through the gas between electrodes (anode and cathode) within the tube.
-
A DC current of 3 to 20 mA is typically required for continue operation.
-
The optical cavityof the laser usually consists of two (fully and partially reflected) concave mirrors or one plane and one concave mirror.
-
The population inversion and light amplification in a He-Ne laser plasma originates with inelastic collision of energetic electrons with ground state helium atoms in the gas mixture.
As shown in energy level diagram, these
-
The collisions excite helium atoms from the ground state to higher energy excited states, among them the \[2{\rm{S}}_1^3\] and \[2{\rm{S}}_0^1\] long-lived metastable states.
-
Because of coincidence between the energy levels of the two He metastable states, and the 3s2 and 2s2 levels of neon, collisions between these helium metastable atoms and ground state neon atoms results in a selective and efficient transfer of excitation energy from the helium to neon.
-
This excitation energy transfer process is given by the reaction equations:
He*( \[2{\rm{S}}_1^3\] ) + Ne \[1{S_0}\] → He( \[1{S_0}\] ) + Ne* \[2{S_2}\] + ΔE
He*( \[2{S^1}\] ) + Ne \[1{S_0}\] + ΔE → He( \[1{S_0}\] ) + Ne* \[3{S_2}\]
-
Where *represents an excited state
-
ΔE is the small energy difference between the energy states of the two atoms. Excitation energy transfer increases the population of the neon \[2{S_2}\] and \[3{S_2}\] levels manyfold.
-
When the population of these two upper levels exceeds that of the corresponding lower level neon state 2P4, to which they are optically connected, population inversion is present.
-
The medium becomes capable of amplifying light in a narrow band at 1.15 μm (corresponding to the \[2{S_2}\] to 2P4 transition) and in a narrow band at 632.8 nm (corresponding to the \[3{S_2}\] to 2P4 transition at 632.8 nm).
-
The 2P4 level is efficiently emptied by fast radiative decay to the 1 \[S\] state, eventually reaching the ground state.
-
The remaining step in utilizing optical amplification to create an optical oscillator is to place highly reflecting mirrors at each end of the amplifying medium.
A stabilized HeNe laser is also one of the benchmark systems for the definition of the meter.[7]
Application
Red He-Ne lasers have many industrial and scientific uses.
They are widely used in laboratory demonstrations in the field of optics.
It produces same quality of spatial coherence and long coherence length at low cost and ease of operation compared to other visible lasers.
Due to visible output, long coherence length and excellent spatial quality it is useful source for holography and as a wavelength reference for spectroscopy.
Prior to the invention of cheap and abundant diode lasers, red He-Ne lasers were widely used in barcode scanners at supermarket checkout count.
He-Ne lasers are generally used in educational and research optical laboratories.
A consumer application of the red He-Ne laser is the Laser Disc player.
The laser is used in the device to read the optical disk.
Ruby Laser
-
Ruby is a crystal of Aluminium Oxide ( \[A{l_2}{O_3}\] ) in which some of the Aluminium ions ( \[Al_3^ +\] ) is replaced by Chromium ions ( \[Cr_3^ +\] ); this is done by doping small amounts of Chromium oxide ( \[C{r_2}{O_3}\] ) in the melt of purified \[A{l_2}{O_3}\]
-
These chromium ions give the crystal a pink or red color depending upon the concentration of chromium ions. It is a solid state laser.
-
Laser rods are prepared from a single crystal of pink ruby which contains 0.05% (by weight) Chromium.
-
\[A{l_2}{O_3}\] does not participate in the laser action. It only acts as the host.
-
The ruby crystal is in the form of cylinder.
-
Length of ruby crystal is usually 2 cm to 30 cm and diameter 0.5 cm to 2 cm.
-
As very high temperature is produced during the operation of the laser, the rod is surrounded by liquid nitrogen to cool the apparatus
Fig17(5 and 6).
Active medium or active center:
-
Chromium ions act as active centers in ruby crystal. So it is the chromium ions that produce the laser.
Pumping source:
-
A helical flash lamp filled with xenon is used as a pumping source.
-
The ruby crystal is placed inside a xenon flash lamp.
-
Thus, optical pumping is used to achieve population inversion in ruby laser.
Optical resonator system:
-
The ends of ruby crystal are polished, grounded and made flat.
-
The one of the ends is completely silvered while the other one is partially silvered to get the output.
-
Thus the two polished ends act as optical resonator system.
Fig 17(5)
Working
-
Ruby is a three level laser system. Suppose there are three levels E1 , E2 and ( E3 & E4).
-
E1 is the ground level, E2 is the metastable level, E3 and E4 are the bands.
-
E3 & E4 are considered as only one level because they are much closed to each other.
Fig 17(6)
Pumping:
-
The ruby crystal is placed inside a xenon flash lamp.
-
The flash lamp is connected to a capacitor which discharges a few thousand joules of energy in a few milliseconds.
-
A part of this energy is absorbed by chromium ions in the ground state. Thus optical pumping raises the chromium ions to energy levels inside the bands E3 and E4 . This process is called stimulated absorption.
-
The transition to bands E3 and E4 are caused by absorption of radiations corresponding to wavelengths approximately 6600 Å and 4000 Å respectively. The levels inside the bands E3 and E4 are also known as pumping levels.
Achievement of population inversion:
-
\[Cr_3^ +\] ions in the excited state loose a part of their energy during interaction with crystal lattice and decay to the metastable state E2 .
-
Thus, the transition from excited states to metastable state is non-radiative transition or in other words there is no emission of photons.
-
As E2 is a metastable state, so chromium ions will stay there for longer time. Hence, the number of chromium ions goes on increasing in E2 state, while due to pumping, the number in the ground state E1 goes on decreasing.
-
As a result, the number of chromium ions becomes more in excited state (metastable state) as compared to ground state E1 .
-
Hence, the population inversion is achieved between states E2 and E1 .
Achievement of laser:
-
Few of the chromium ions will come back from E2 to E1 by the process of spontaneous emission by emitting photons (6943Å ).
-
This photon travels through the ruby rod and if it is moving in a direction parallel to the axis of the crystal, then it is reflected to and fro by the silvered ends of the ruby rod until it stimulates the other excited ions and cause it to emit a fresh photon in phase with the stimulating photon.
-
Thus, the reflections will result in stimulated emission and it will result in the amplification of the stimulated emitting photons.
-
This stimulated emission is the laser transition.
-
The two stimulated emitted photons will knock out more photons by stimulating the chromium ions and their total number will be four and so on.
-
This process is repeated again and again, thus photons multiply. When the photon beam become sufficiently intense, then a very powerful and narrow beam of red light of wavelength 6943 Å emerges through the partially silvered end of the ruby crystal.
-
In the energy level diagram, E2 is the upper laser level and E1 is the lower laser level because laser beam is achieved in between these levels. Thus, the ruby laser fits into the definition of three level laser systems.
Drawbacks of ruby laser
-
As the terminus of laser action is the ground state, it is difficult to maintain the population inversion which gives low efficiency.
-
The ruby laser requires high power pumping source.
-
Laser output is not continuous but occurs in the form of pulses ( \[\mu s\] ).
-
The defects due to crystalline imperfection are also present in ruby laser.
Uses of ruby laser
-
Ruby laser has very high output power of the order of 104 - 106 watts.
-
It has wavelength of 6943 Å .
-
Ruby lasers are used for holography, industrial cutting and welding.